
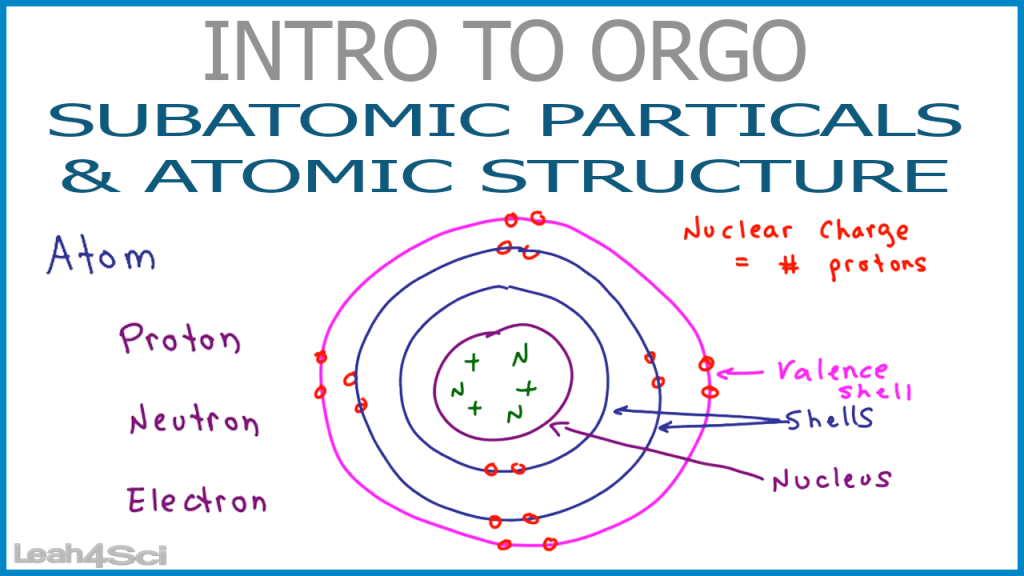
Because opposite charges attract each other (while like charges repel each other), protons attract electrons (and vice versa).įinally, additional experiments pointed to the existence of a third particle. Although still incredibly small, the mass of a proton is 1.673 × 10 −27 kg, which is almost 2,000 times greater than the mass of an electron. Another major difference between a proton and an electron is mass. 
has the same amount of charge as an electron, but its charge is positive, not negative. A proton A subatomic particle with a positive charge. Experiments with magnetic fields showed that the electron has a negative electrical charge.īy 1920, experimental evidence indicated the existence of a second particle. It is an extremely tiny particle, with a mass of about 9.109 × 10 −31 kg. For example in carbon – 14, there are 6 protons and 8 neutrons and thus it shows radioactivity.The first subatomic particle was identified in 1897 and called the electron A subatomic particle with a negative electric charge. An element becomes unstable when the number of neutrons in it is abnormally high. 
Neutrons produce gamma rays during radioactivity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
